| Journal of Hematology, ISSN 1927-1212 print, 1927-1220 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Hematol and Elmer Press Inc |
| Journal website https://www.thejh.org |
Case Report
Volume 11, Number 1, February 2022, pages 15-20
Leukoerythroblastosis in a Sickle Cell Patient With Pregnancy: An Interesting Peripheral Blood Smear Finding
Anupam Mitraa, b, Ananya Datta Mitraa, b, Gopal Patela, Denis M. Dwyrea, John P. Graffa, c
aDepartment of Pathology and Laboratory Medicine, University of California Davis, School of Medicine, 4400 V Street, PATH Building, Sacramento, CA 95817, USA
bThese authors equally contributed to the work as joint first authors.
cCorresponding Author: John P. Graff, Department of Pathology and Laboratory Medicine, University of California Davis School of Medicine, 4400 V Street, PATH Building, Sacramento, CA 95817, USA
Manuscript submitted September 9, 2021, accepted October 21, 2021, published online December 13, 2021
Short title: Leukoerythroblastosis in a Pregnant SCD Patient
doi: https://doi.org/10.14740/jh921
| Abstract | ▴Top |
The global pandemic of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has shaken the entire world. The social, health and financial impacts of this pandemic are beyond words. We have learnt a lot about this new disease in a short period of time, but still a long road to go to fully determine its pathogenic effect. The primary target of this virus is angiotensin-converting enzyme 2 (ACE2) receptor, which is prevalent in endothelial cells throughout the body. Immunocompromised patients such as patients with sickle cell disease are more vulnerable to severe respiratory infections, including infection with SARS-CoV-2. In addition, sickle cell disease patients are prone to vaso-occlusive crisis, and theoretically SARS-CoV-2 can worsen the situation as it also can cause endothelial dysfunction and thrombosis. Herein, we are sharing an interesting peripheral blood smear finding of an asymptomatic 31-year-old multigravida pregnant female with a history of sickle cell disease and found to have a positive COVID-19 polymerase chain reaction (PCR) test during her third trimester of pregnancy at a routine clinic visit. Two weeks after the initial positive test, she developed nausea, vomiting, constipation and a pain crisis affecting her extremities while her COVID-19 PCR test was still positive. She was hemodynamically stable, and lab workup revealed chronic anemia, leukocytosis with neutrophilia and lymphopenia. Morphologic examination of the peripheral blood smear showed a marked leukoerythroblastosis: rare myeloblasts, sickle cells, markedly abundant nucleated red blood cells (RBCs), metamyelocytes, and many large and giant platelets were seen. In this context, her previous peripheral blood smears (prior to positive COVID-19 test) did not show leukoerythroblastosis. She was managed conservatively with hydration and pain control and delivered at 36 weeks via cesarean section due to pre-term labor and intrauterine growth retardation. The unusual finding of leukoerythroblastosis in a pregnant sickle cell disease patient with an asymptomatic COVID-19 infection indicates further studies to determine its effect on hematopoietic system and elucidate its clinical significance.
Keywords: COVID-19; Sickle cell disease; Pregnancy; Leukoerythroblastosis
| Introduction | ▴Top |
The recent outbreak of novel coronavirus infection, which was first reported in China, is responsible for the global pandemic known as coronavirus disease 2019 (COVID-19). This disease is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), spreads via respiratory droplets, and is characterized by a spectrum of manifestations ranging from asymptomatic or mild flu-like symptoms to severe respiratory failure and death. People with chronic illnesses, compromised immune system, and elderly patients are more vulnerable to this infection with increased morbidity and mortality. Literature suggests that SARS-CoV-2 causes more severe respiratory symptoms in immunocompromised patients as well it causes endothelial dysfunction and thrombosis [1-4]. The adverse prognostic factors include advanced age, immunosuppression, obesity, diabetes, cardiovascular, pulmonological renal diseases, pregnancy, and sickle cell disease (SCD) [1, 3, 5, 6].
In the USA, African Americans are disproportionately affected by SARS-CoV-2 more than any other racial groups [7]. SCD is one of the most common hemoglobinopathies in the world and people with the disease are more prone to SARS-CoV-2 infection due to their immunosuppressive state from the underlying disease and/or therapies [8, 9]. If infected, these patients have an increased risk of developing severe respiratory complications, including acute respiratory distress syndrome (ARDS), pneumonia and secondary bacterial infections [8]. It has been reported that morbidity and mortality of SARS-CoV-2 is disproportionately higher among African Americans with the exact cause being unknown [10, 11]. SCD is also more common in this patient population [12, 13]. SCD patients are immunologically challenged and more vulnerable to manifest severe respiratory infection and vaso-occlusive symptoms from SARS-CoV-2 infection [14].
So far published data from different parts of the world on SARS-COV-2 and SCD patients suggest conflicting outcomes. A few studies from Europe and the USA suggest that SARS-CoV-2 does not increase the mortality in SCD patients [8, 9, 15], whereas another study suggests detrimental effects of SARS-CoV-2 infection in sickle cell patients [16]. The current data for ideal management of SCD patients presenting with SARS-CoV-2 are still under evaluation due to the scarcity of published data.
Pregnancy causes a transient immunocompromised state in the mother to protect the fetus, which increases mother’s susceptibility to various respiratory pathogens [17]. Studies have shown that angiotensin-converting enzyme 2 (ACE2) receptor on host cell surface in lung, erythroid, endothelial cells, and placenta is responsible for mediating SARS-CoV-2 infection in humans [18, 19]. Being immunocompromised and with the presence of ACE2 receptors in the placenta increases the risk of SARS-CoV-2 infection during pregnancy [18, 20]. A few case series have shown that pregnant patients with SARS-CoV-2 have presented with fever, pneumonia, cough and dyspnea, similar to that of non-pregnant patients with SARS-CoV-2 infection. With this presentation, many of these patients deliver via cesarean section [18, 21]. Very few of these patients required admission in the intensive care unit and mechanical ventilation, and there was no maternal mortality reported [18]. The most common symptoms of SARS-CoV-2 in these patients are fever, dry cough, dyspnea, and myalgias, whereas hematological complications include lymphopenia, thrombocytopenia, coagulation disorders, and thrombotic events [1, 2, 5, 6, 22].
SCD patients are immunocompromised and are prone to develop thrombosis. Pregnancy is also known to cause some degree of physiological immunosuppression and an overall pro-thrombotic state. Thus, pregnancy in SCD patients makes them more vulnerable to infection, including SARS-CoV-2, and thrombotic episodes. SARS-CoV-2 is found to cause endothelial dysfunction and thrombosis. Thus, pregnancy in a SCD patient with SARS-CoV-2 infection may have an increased risk of adverse outcomes. To date, there are no published reports on this topic. Here, we are presenting an interesting case of SARS-CoV-2 infection in a pregnant SCD patient. Surprisingly, the outcome of this case was not severe; however we noticed an unusual presentation of leukoerythroblastosis in her peripheral blood smear.
| Case Report | ▴Top |
Investigations
An asymptomatic pregnant 31-year-old female (G6P4) was found to have SARS-CoV-2 infection during routine nasopharyngeal swab testing at 30 weeks of gestation. She also has a history of SCD (HbSS). At 32 weeks of gestation, she presented with nausea, vomiting, constipation, decreased oral intake and a pain crisis limited to her extremities. She continued to test positive for SARS-CoV-2 on repeat testing. She denied fever, shortness of breath or respiratory distress, chest pain or cough. Although these are nonspecific features of pulmonary SARS-CoV-2, the presence of these symptoms may have complicated her clinical picture as to whether they are caused by her infection or due to acute chest syndrome from her SCD. On examination, she was in moderate distress due to pain, but hemodynamically stable and was maintaining her oxygen saturation. Clinical examination of other systems was within normal limit. Ultrasound of the abdomen showed a normal viable fetus with normal heart rate and accelerations.
Diagnosis
Her laboratory values during this hospital stay are shown in Table 1. Briefly, her complete blood count (CBC) showed leukocytosis (white blood cell (WBC): 15.9 × 109/L) with neutrophilia (87.5%), lymphopenia (7.7%) and normal monocyte count (4.8%). She has chronic normocytic anemia (hemoglobin: 8.9 g/dL, mean corpuscular volume (MCV): 99.8), presence of a moderate number of sickle cells with a normal platelet count (335 × 109/L). Viral panels (cytomegalovirus (CMV), parvovirus B19 and Epstein-Barr virus (EBV)) were undetectable for virus by polymerase chain reaction (PCR) testing. SARS-CoV-2 was detected on two occasions by PCR (Table 2).
 Click to view | Table 1. Complete Blood Count With Differential |
 Click to view | Table 2. Results for SARS-CoV-2 (PCR) |
Treatment
The patient was managed conservatively with intravenous hydration and pain medications and was discharged 5 days after admission.
Follow-up and outcomes
She returned to the emergency department the following day with increasing pain in her extremities. Her CBC was notable for persistent leukocytosis (WBC: 20.4 × 109/L) with neutrophilia (75.7%), lymphopenia (16.5%) and a normal monocyte count (6.8%). Morphologic examination of the peripheral blood smear showed the presence of a moderate number of sickle cells and marked leukoerythroblastosis: rare myeloblasts (about 1%), markedly abundant nucleated red blood cells (RBCs) (32 nucleated RBCs per 100 WBCs), 1.0% metamyelocytes, and many large and giant platelets were seen (Fig. 1). Her previous peripheral blood smears (prior to the positive SARS-CoV-2 test) did not show leukoerythroblastosis. There was a rise in the percentage of HbS during her pregnancy, and she received a red cell transfusion (Table 3). She received intravenous hydration and pain medications until her delivery. The baby was delivered at 36 weeks via cesarean section due to pre-term labor and intrauterine growth retardation. The leukoerythroblastic picture was transient and was not appreciated 2 weeks post-delivery. The placenta of the patient was histologically mature but small for gestation age and had a three-vessel umbilical cord. No significant pathological changes were noted in the placenta.
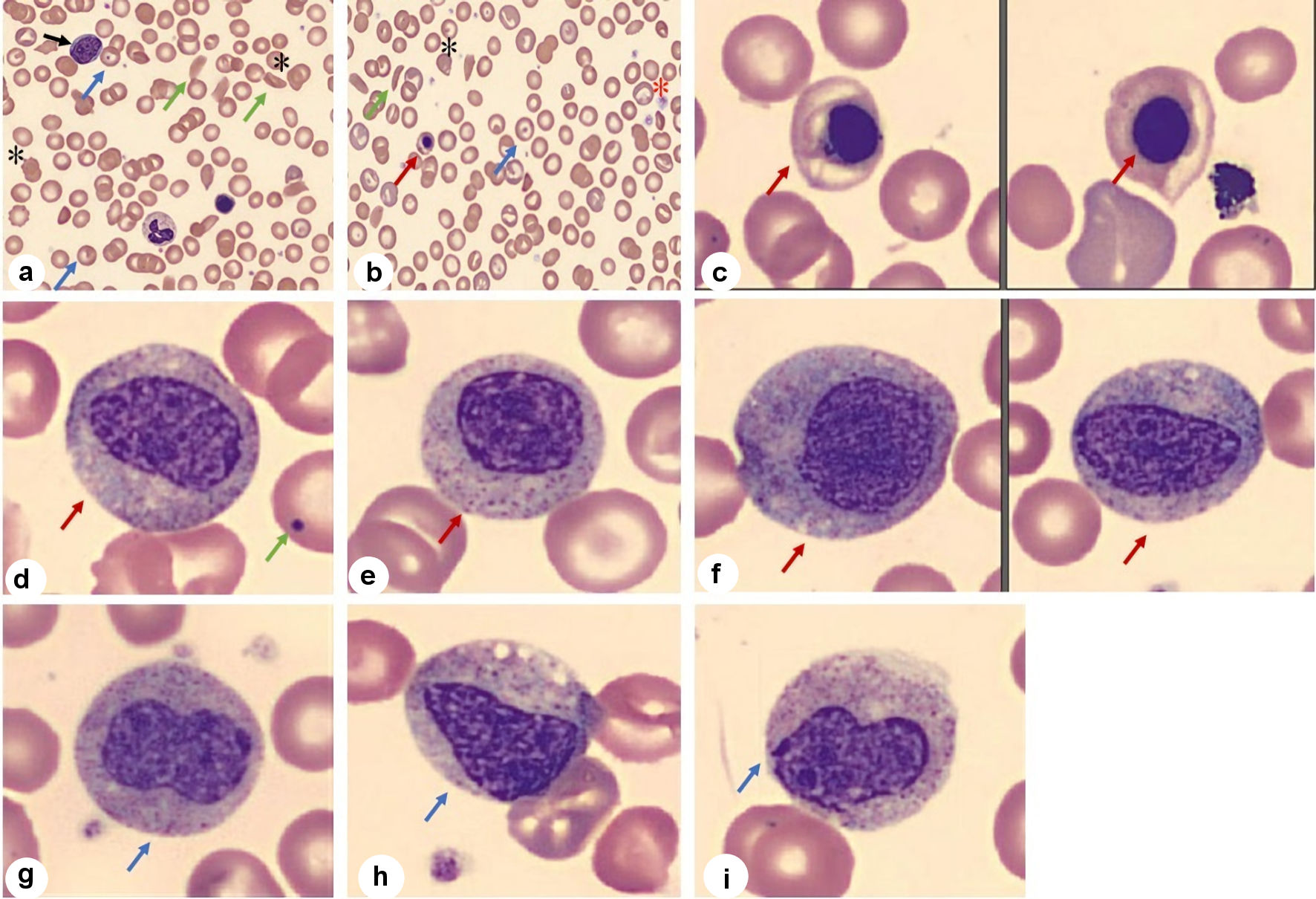 Click for large image | Figure 1. Peripheral blood smear. (a-c) Smear shows presence of abnormal RBC morphology in low and high magnification: sickle cells (green arrow), target cells (blue arrow), schistocytes (*), nucleated RBCs (red arrow) along with giant platelets (*) and myeloblast (black arrow). (d-f) Smear shows presence of myelocytes (red arrow) with Howell-Jolly body (green arrow) (d). (g-i) Smear shows metamyelocytes (blue arrow). RBC: red blood cell. |
 Click to view | Table 3. Hemoglobin Electrophoresis Results |
| Discussion | ▴Top |
The peripheral blood smear of our SCD patient showed immature erythroid and myeloid cells, including rare myeloblasts, during her SARS-CoV-2 infection. Such findings can be concerning for a myeloproliferative neoplasm (MPN) in other clinical settings, but its implication is unknown in SARS-CoV-2 patients. This case was diagnosed with transient leukoerythroblastosis in the setting of an acute SARS-CoV-2 infection. It has been shown by a few groups, including ours, that immature erythroid and myeloid precursors are seen in peripheral smear of patients with SARS-CoV-2 infection [23, 24]. Based on a current literature search, we believe that our case is the first study to report leukoerythroblastosis in a pregnant SCD patient with SARS-CoV-2 infection.
Leukoerythroblastosis is defined by the presence of immature erythroid and myeloid precursors in peripheral circulation. It can also be seen with certain infections, such as parvovirus [25-28] and malaria [29], and in metastatic carcinomas [30-32]. We do not know the clinical significance of this finding in COVID-19, but we are hopeful that this observation will guide us to understand its impact in the near future. There are reports suggesting that SARS-CoV-2 infection can induce pathological changes in placenta, such as decidual arteriopathy, features of maternal vascular malperfusion (presence of intervillous thrombi), and pre-eclampsia like syndrome [33, 34], but we did not see any of these pathological changes in our patient.
Leukoerythroblastosis is not a feature of SCD. Pregnancy in SCD patients causes some pathophysiologic changes to meet the demand of pregnancy, but leukoerythroblastosis in not one of them. SCD patients are pro-thrombotic and immunocompromised, and pregnancy also causes further immunosuppression and accentuates the pro-thrombotic state. Thus, pregnancy in SCD patients requires more intense care to meet the clinical demand and is often categorized as a high-risk group. Prior published reports suggest that SARS-CoV-2 infections can cause more severe respiratory infection symptoms in immunocompromised patients and induces endothelial dysfunction and thrombosis. Thus, the rational expectation is that SARS-CoV-2 infection might play additive or synergistic role and make the clinical outcome worse in SCD patients while pregnant. Published data from different parts of the world indicate that clinical outcome is not more severe in most cases of SARS-CoV-2 infection in patients with SCD [8, 9, 15, 16]. One possible explanation for this clinical outcome might be that most of the patients with SCD are relatively younger and receive more frequent medical attention for their underlying disease. Published studies suggest that pregnant women with SARS-CoV-2 infections have more cesarean sections and more placental pathological abnormalities, but with no increased maternal mortality reported to date [18, 20, 33-35].
SCD patients are at increased risk of acute pulmonary illness and viral infections [36]. Previous studies have shown that patients with SCD have increased rates of hospitalization and intensive care admission when infected with seasonal influenza [37]. Furthermore, these patients are also at increased risk of vaso-occlusive crises and acute chest syndrome with hypoxia induced by the infection and is a major cause of mortality in these patients [38]. A case series from India [39] also showed an increased risk of pregnancy complications in SCD patients with COVID-19. They noted in their cohort that this increased risk might not be related to the anemia or respiratory issues in SCD and COVID-19. Similarly, these patients are more prone to severe SARS-CoV-2 infection related complications than the normal population as their immune system is compromised by the disease process as well as by therapy (hydroxyurea) [14, 39]. This is an open and active area of research.
As the leukoerythroblastic peripheral blood findings seen in this patient were transient, there was little clinical concern for a primary bone marrow disease or bone marrow necrosis, and therefore a bone marrow biopsy was not indicated. Bone marrow biopsy findings in SCD patients with leukoerythroblastosis have been reported. These studies showed evidence of bone marrow necrosis in their marrow [40]. Focal bone marrow necrosis can be a complication of vaso-occlusive crises in SCD patients. They can present with a leukoerythroblastic blood picture. In the reported case series, two patients had pancytopenia, high fevers, and one had a strikingly high nucleated RBC count.
In this context, our group and others have recently showed leukoerythroblastic peripheral smear in a COVID-19 patient without any history of underlying hematological malignancy [23].
Learning point
This case is suggesting that the clinical outcome of SARS-CoV-2 infection in a pregnant SCD patient is not as worrisome as we thought. This finding is in line with other published reports [18, 21]. The unusual presence of leukoerythroblastosis in the peripheral smear suggests the importance of reviewing the peripheral smear in COVID-19 patients, more so than with asymptomatic patients. We believe that this observation will help the scientific community to understand the pathogenic impact of this coronavirus on hematopoietic system. This finding might be served as a surrogate marker depending on future studies. More studies are needed to elucidate the clinical significance of this finding.
Acknowledgments
We acknowledge Dr. Ted Wun (hemato-oncologist) and his clinical team’s effort to get the consent from this patient. We acknowledge Dr. Anthony Karnezis (gynecology pathologist) for providing placental findings. We also appreciate patient’s cooperation to share this finding among medical community.
Financial Disclosure
None to declare.
Conflict of Interest
The authors declare that they have no competing interest.
Informed Consent
We received signed informed consent from the patient to publish this finding.
Author Contributions
AM and ADM looked into the case details and drafted the manuscript. GP, JPG and DD edited the manuscript and supervised the work.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
ARDS: acute respiratory distress syndrome; MPN: myeloproliferative neoplasm; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; SCD: sickle cell disease; WBC: white blood cell
| References | ▴Top |
- Bhatraju PK, Ghassemieh BJ, Nichols M, Kim R, Jerome KR, Nalla AK, Greninger AL, et al. Covid-19 in critically ill patients in the Seattle region - case series. N Engl J Med. 2020;382(21):2012-2022.
doi pubmed - Klok FA, Kruip M, van der Meer NJM, Arbous MS, Gommers D, Kant KM, Kaptein FHJ, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145-147.
doi pubmed - Onder G, Rezza G, Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA. 2020;323(18):1775-1776.
doi pubmed - Panigada M, Bottino N, Tagliabue P, Grasselli G, Novembrino C, Chantarangkul V, Pesenti A, et al. Hypercoagulability of COVID-19 patients in intensive care unit: A report of thromboelastography findings and other parameters of hemostasis. J Thromb Haemost. 2020;18(7):1738-1742.
doi pubmed - Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323(11):1061-1069.
doi pubmed - Guan WJ, Liang WH, Zhao Y, Liang HR, Chen ZS, Li YM, Liu XQ, et al. Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis. Eur Respir J. 2020;55(5):2001227.
doi pubmed - Gupta R, Agrawal R, Bukhari Z, Jabbar A, Wang D, Diks J, Alshal M, et al. Higher comorbidities and early death in hospitalized African-American patients with Covid-19. BMC Infect Dis. 2021;21(1):78.
doi pubmed - Arlet JB, de Luna G, Khimoud D, Odievre MH, de Montalembert M, Joseph L, Chantalat-Auger C, et al. Prognosis of patients with sickle cell disease and COVID-19: a French experience. Lancet Haematol. 2020;7(9):e632-e634.
doi - McCloskey KA, Meenan J, Hall R, Tsitsikas DA. COVID-19 infection and sickle cell disease: a UK centre experience. Br J Haematol. 2020;190(2):e57-e58.
doi pubmed - Yancy CW. COVID-19 and African Americans. JAMA. 2020;323(19):1891-1892.
doi pubmed - Fouad MN, Ruffin J, Vickers SM. COVID-19 Is disproportionately high in African Americans. This will come as no surprise. Am J Med. 2020;133(10):e544-e545.
doi pubmed - Wastnedge E, Waters D, Patel S, Morrison K, Goh MY, Adeloye D, Rudan I. The global burden of sickle cell disease in children under five years of age: a systematic review and meta-analysis. J Glob Health. 2018;8(2):021103.
doi pubmed - Hassell KL. Population estimates of sickle cell disease in the U.S. Am J Prev Med. 2010;38(4 Suppl):S512-521.
doi pubmed - Kehinde TA, Osundiji MA. Sickle cell trait and the potential risk of severe coronavirus disease 2019-A mini-review. Eur J Haematol. 2020;105(5):519-523.
doi pubmed - Balanchivadze N, Kudirka AA, Askar S, Almadhoun K, Kuriakose P, Fadel R, Dabak V. Impact of COVID-19 infection on 24 patients with sickle cell disease. One Center Urban Experience, Detroit, MI, USA. Hemoglobin. 2020;44(4):284-289.
doi pubmed - Panepinto JA, Brandow A, Mucalo L, Yusuf F, Singh A, Taylor B, Woods K, et al. Coronavirus disease among persons with sickle cell disease, United States, March 20-May 21, 2020. Emerg Infect Dis. 2020;26(10):2473-2476.
doi pubmed - Poole JA, Claman HN. Immunology of pregnancy. Implications for the mother. Clin Rev Allergy Immunol. 2004;26(3):161-170.
doi - Dashraath P, Wong JLJ, Lim MXK, Lim LM, Li S, Biswas A, Choolani M, et al. Coronavirus disease 2019 (COVID-19) pandemic and pregnancy. Am J Obstet Gynecol. 2020;222(6):521-531.
doi pubmed - Shahbaz S, Xu L, Osman M, Sligl W, Shields J, Joyce M, Tyrrell DL, et al. Erythroid precursors and progenitors suppress adaptive immunity and get invaded by SARS-CoV-2. Stem Cell Reports. 2021;16(5):1165-1181.
doi pubmed - Phoswa WN, Khaliq OP. Is pregnancy a risk factor of COVID-19? Eur J Obstet Gynecol Reprod Biol. 2020;252:605-609.
doi pubmed - Della Gatta AN, Rizzo R, Pilu G, Simonazzi G. Coronavirus disease 2019 during pregnancy: a systematic review of reported cases. Am J Obstet Gynecol. 2020;223(1):36-41.
doi pubmed - Zhang Y, Xiao M, Zhang S, Xia P, Cao W, Jiang W, Chen H, et al. Coagulopathy and antiphospholipid antibodies in patients with COVID-19. N Engl J Med. 2020;382(17):e38.
doi pubmed - Mitra A, Dwyre DM, Schivo M, Thompson GR, 3rd, Cohen SH, Ku N, Graff JP. Leukoerythroblastic reaction in a patient with COVID-19 infection. Am J Hematol. 2020;95(8):999-1000.
doi pubmed - Lee WS, Margolskee E. Leukoerythroblastosis and plasmacytoid lymphocytes in a child with SARS-CoV-2-associated multisystem inflammatory syndrome. Blood. 2020;136(7):914.
doi pubmed - Ozdemir N, Aki H, Hakyemez HT, Cokugras FC, Apak H. Parvovirus B19 infection mimicking juvenile myelomonocytic leukemia. Int J Infect Dis. 2010;14(Suppl 3):e379-380.
doi pubmed - Anand A, Gray ES, Brown T, Clewley JP, Cohen BJ. Human parvovirus infection in pregnancy and hydrops fetalis. N Engl J Med. 1987;316(4):183-186.
doi pubmed - Gulen H, Basarir F, Hakan N, Ciftdogan DY, Tansug N, Onag A. Premature labor and leukoerythroblastosis in a newborn with parvovirus B19 infection. Haematologica. 2005;90(Suppl):ECR38.
- Patel PA, Weinzierl EP, Wechsler DS. Leukoerythroblastosis as an unusual presentation of parvovirus B19 infection in a sickle cell patient. Case Rep Pediatr. 2020;2020:8841607.
doi pubmed - Varo R, Sitoe A, Cossa A, Ordi J, Rozman M, Bassat Q. Leukoerythroblastosis in a young child with severe malaria and superimposed gram negative infection. J Trop Pediatr. 2018;64(6):553-556.
doi pubmed - Delsol G, Guiu-Godfrin B, Guiu M, Pris J, Corberand J, Fabre J. Leukoerythroblastosis and cancer frequency, prognosis, and physiopathologic significance. Cancer. 1979;44(3):1009-1013.
doi - Fan FS, Yang CF. Leukoerythroblastosis in castration-resistant prostate cancer: A clue to diffuse bone marrow carcinomatosis. Clin Pract. 2019;9(2):1124.
doi pubmed - Canbolat Ayhan A, Timur C, Ayhan Y, Kes G. Leukoerythroblastosis mimicking leukemia: a case report. Iran J Pediatr. 2014;24(3):332-333.
doi pubmed - Mendoza M, Garcia-Ruiz I, Maiz N, Rodo C, Garcia-Manau P, Serrano B, Lopez-Martinez RM, et al. Pre-eclampsia-like syndrome induced by severe COVID-19: a prospective observational study. BJOG. 2020;127(11):1374-1380.
doi pubmed - Shanes ED, Mithal LB, Otero S, Azad HA, Miller ES, Goldstein JA. Placental pathology in COVID-19. Am J Clin Pathol. 2020;154(1):23-32.
doi pubmed - Narang K, Enninga EAL, Gunaratne M, Ibirogba ER, Trad ATA, Elrefaei A, Theiler RN, et al. SARS-CoV-2 infection and COVID-19 during pregnancy: a multidisciplinary review. Mayo Clin Proc. 2020;95(8):1750-1765.
doi pubmed - Siddiqui AK, Ahmed S. Pulmonary manifestations of sickle cell disease. Postgrad Med J. 2003;79(933):384-390.
doi pubmed - Bundy DG, Strouse JJ, Casella JF, Miller MR. Burden of influenza-related hospitalizations among children with sickle cell disease. Pediatrics. 2010;125(2):234-243.
doi pubmed - Vichinsky EP, Neumayr LD, Earles AN, Williams R, Lennette ET, Dean D, Nickerson B, et al. Causes and outcomes of the acute chest syndrome in sickle cell disease. National Acute Chest Syndrome Study Group. N Engl J Med. 2000;342(25):1855-1865.
doi pubmed - Waghmare R, Chaaithanya IK, Zala S, Deshmukh J, Uikey P, Wankhede S, Palve T, et al. Outcomes of COVID-19 in pregnant women with sickle cell disease in India: a case series. Indian J Hematol Blood Transfus. 2021:1-3.
doi pubmed - Lederman HM, Connolly MA, Kalpatthi R, Ware RE, Wang WC, Luchtman-Jones L, Waclawiw M, et al. Immunologic effects of hydroxyurea in sickle cell anemia. Pediatrics. 2014;134(4):686-695.
doi pubmed - Ataga KI, Orringer EP. Bone marrow necrosis in sickle cell disease: a description of three cases and a review of the literature. Am J Med Sci. 2000;320(5):342-347.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Hematology is published by Elmer Press Inc.


